What are the derivatives of Phenylboronic Acid?
Leave a message
Phenylboronic acid is a versatile compound that finds numerous applications in organic synthesis, medicinal chemistry, and materials science. As a leading supplier of phenylboronic acid and its derivatives, we are committed to providing high-quality products to meet the diverse needs of our customers. In this blog post, we will explore the various derivatives of phenylboronic acid and their significance in different fields.
Introduction to Phenylboronic Acid
Phenylboronic acid has the chemical formula C₆H₅B(OH)₂. It consists of a phenyl group (C₆H₅) attached to a boronic acid functional group (-B(OH)₂). The boronic acid group can undergo a variety of reactions due to the unique properties of boron. It can form reversible covalent bonds with diols and other Lewis bases, which makes it useful in a wide range of chemical transformations.
Common Derivatives of Phenylboronic Acid
Halogen - Substituted Phenylboronic Acids
Halogen - substituted phenylboronic acids are important derivatives. For example, 4 - bromophenylboronic acid and 3 - chlorophenylboronic acid. These compounds are widely used in cross - coupling reactions, such as the Suzuki - Miyaura reaction. The Suzuki - Miyaura reaction is a palladium - catalyzed coupling of an organoboron compound (in this case, a phenylboronic acid derivative) with an organohalide or a pseudo - halide. It has become one of the most powerful methods for the construction of carbon - carbon bonds in organic synthesis.
The presence of a halogen atom on the phenyl ring can introduce additional reactivity or selectivity in the reaction. For instance, 4 - bromophenylboronic acid can react with various aryl halides to form biaryl compounds, which are important structural motifs in many natural products, pharmaceuticals, and functional materials.

Alkyl - Substituted Phenylboronic Acids
Alkyl - substituted phenylboronic acids, like 4 - methylphenylboronic acid and 3 - ethylphenylboronic acid, are also frequently used. The alkyl groups can affect the electronic and steric properties of the phenylboronic acid. Electron - donating alkyl groups can increase the electron density on the phenyl ring, which may influence the reactivity in certain reactions.
These derivatives are used in the synthesis of complex organic molecules. For example, in the preparation of agrochemicals, the introduction of an alkyl - substituted phenylboronic acid derivative can fine - tune the biological activity and solubility of the final product.
Heteroatom - Substituted Phenylboronic Acids
Phenylboronic acid derivatives with heteroatoms such as nitrogen, oxygen, and sulfur on the phenyl ring are also of great interest. For example, 4 - (dimethylamino)phenylboronic acid contains a nitrogen - containing functional group. This type of compound can participate in reactions where the nitrogen lone pair can interact with other reagents, leading to unique reaction pathways.
Oxygen - substituted derivatives like 4 - methoxyphenylboronic acid are commonly used in the synthesis of pharmaceuticals and natural product mimics. The methoxy group can act as an electron - donating group and also affect the solubility and lipophilicity of the compound.
Applications of Phenylboronic Acid Derivatives in Medicinal Chemistry
Phenylboronic acid derivatives play a crucial role in medicinal chemistry. They are used in the discovery and development of new drugs. For example, some phenylboronic acid - based compounds have been developed as protease inhibitors. The boronic acid group can form a covalent bond with the active site of the protease, inhibiting its enzymatic activity.
In addition, Semaglutide CAS 910463 - 68 - 2 is an important peptide - based drug. Phenylboronic acid derivatives may be used in the synthesis of its intermediates. The unique properties of phenylboronic acid can help in the construction of complex molecular structures in a controlled manner during the drug synthesis process.
Applications in Materials Science
In materials science, phenylboronic acid derivatives are used in the preparation of functional polymers and organic thin - film materials. For example, they can be incorporated into polymers to impart specific properties such as self - healing ability. The reversible covalent bonds formed by the boronic acid group can allow the polymer to repair itself when damaged.
Phenylboronic acid derivatives can also be used in the preparation of organic light - emitting diodes (OLEDs). They can act as charge - transporting materials or emitters, improving the performance of the OLED devices.
Applications in Organic Synthesis
As mentioned earlier, phenylboronic acid derivatives are key reagents in cross - coupling reactions. The Suzuki - Miyaura reaction has been widely used in the synthesis of natural products, pharmaceuticals, and advanced materials.
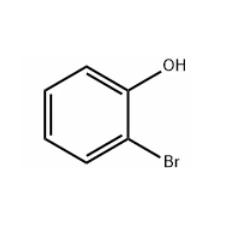
For example, 2 - bromophenol CAS 95 - 56 - 7 can react with an appropriate phenylboronic acid derivative in the presence of a palladium catalyst to form a substituted phenol product. This reaction is highly selective and efficient, allowing chemists to construct complex molecules with high precision.
Other Related Compounds and References
In addition to the above - mentioned applications, our company also supplies other related compounds. For example, High Purity Reagent Succinimidyl Myristate (SM) CAS 69888 - 86 - 4, which is an important pharmaceutical intermediate.
If you are interested in our phenylboronic acid derivatives or other products, please feel free to contact us for procurement and negotiation. We are dedicated to providing you with top - quality products and excellent service.
References
- Miyaura, N.; Suzuki, A. Palladium - catalyzed cross - coupling reactions of organoboron compounds. Chem. Rev. 1995, 95, 2457 - 2483.
- Hall, D. G. Boronic Acids: Preparation and Applications in Organic Synthesis, Medicine and Materials. Wiley - VCH, 2011.
- Hartwig, J. F. Organotransition Metal Chemistry: From Bonding to Catalysis. University Science Books, 2010.